We are pursuing promising “hits” for novel antibiotics active against Gram–negative bacteria and mechanism-based therapeutics for diabetes and obesity.
The current research effort in our laboratory is focused on two proteins. The first involves the critical digestive enzyme human pancreatic alpha-amylase, with the immediate goals of determining the catalytic mechanism of this enzyme and how substrates bind in the active site region next to catalytic residues. As part of this process our laboratory has solved the full three-dimensional atomic structure of human pancreatic alpha-amylase (see drawing below). This structural information, in combination with other functional studies, is being used as the starting point for the design of inhibitors to act as therapeutic drug agents in the treatment of diabetes, obesity and dental caries.
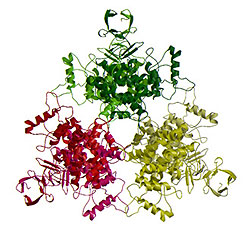
A second system being examined involves a unique hexameric form of the enzyme citrate synthase, which is only found in Gram-negative bacteria. This key metabolic enzyme is allosterically controlled by an unknown mechanism. Our laboratory has recently solved the complete three-dimensional structure of this enzyme (see drawing below) and begun the process of unraveling the structure-function relationships that control its catalytic activity. Given the central role this enzyme plays in the metabolism of Gram-negative pathogens, understanding its mechanism of action could potentially allow for the development of novel anti-microbial compounds.